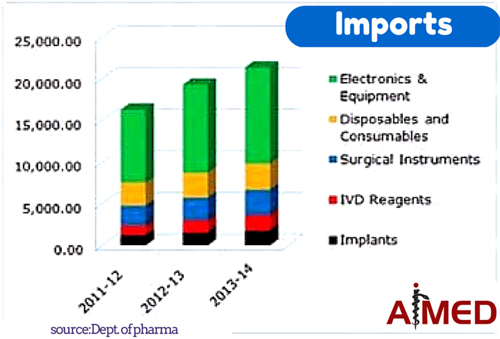
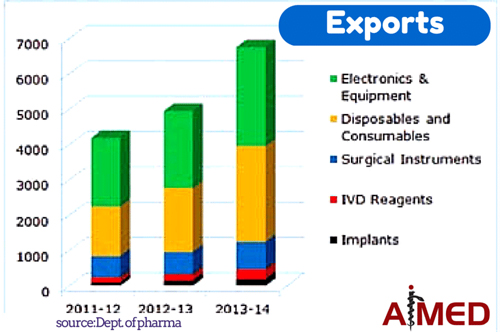
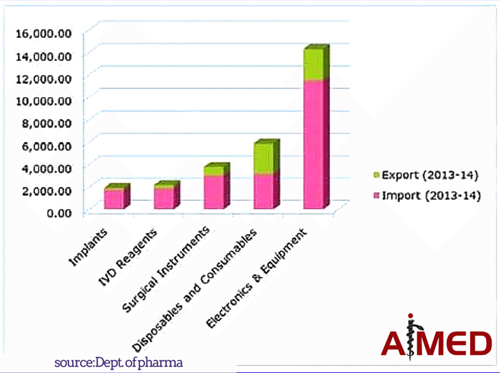
1. VIABILITY
Investors Invest if business is viable &
profitable
Correct Import Duties
• Withdraw all Concessional Duty Notifications
• Revert Basic Duty from 0 or 5% to 10%
• Revert SAD from 0% to 4%
2. BUY INDIAN POLICY
Preferential Market Access & Pricing Policy
Public Health Procurement
• Preferential Market Access
• 15% preferential pricing policy of Indian Origin
Devices
• 5% preferential pricing for IS:13485 certified
manufacturers
3. CONSUMER PROTECTION
Control Artificial Inflation (of MRP)
Control Artificial Inflation
|
• Enforce MRP on unit pack
• Impose 1% Excise Duty on MRP
• Impose Excise Duty on MRP less, over 50% abatement
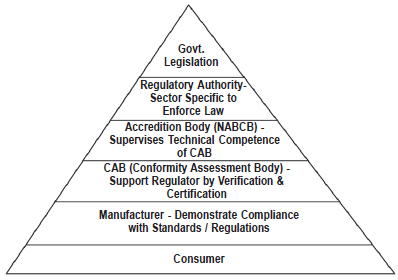
4. REGULATIONS
Incorrect Regulations discourage Investments
Regulatory Framework
• Separate Law Book
• Separate Rule Book
• Traders not be permitted to be called as
Manufacturers
• Separate Regulatory Body or Revamped CDSCO
• IHPRA - Indian Healthcare Products Regulatory
Authority
• 3rd Party Certification
Self Regulations
• AIMED & QCI Initiative
• Voluntary Certification
• IC - MED 13485 Scheme
5. FOREIGN DIRECT INVESTMENTS
100% FDI restricted to Greenfield Manufacturing
Restrict 100% FDI to Green Field
• In a Manufacturing (do not permit for trading)
• At least 60% Turnover from Manufacturing
• Ban Auto Route For Buying Indian Manufacturers
6. DEPARTMENT OF MEDICAL DEVICES
Coordinator & Nodal Interface - Facilitator
MINISTRY OF
HEALTHCARE PRODUCTS
7. PRODUCT DEVELOPMENT
Incentivise Innovation
Commercialising R&D
• Forum for USERS, Developers & Manufacturers
• Commercialization not Academic Acclaim
• Bringing in Utility model, Expediting Patent
Application processing
• Create SPV to own patents/IP infuse 100 million$
equity for R&D projects
THE VISION :
INDIA AMONGST TOP 5
MANUFACTURING HUBS OF
MEDICAL DEVICES
 |
